The TMAH experimental cup consisted of two sealed foil caps. The outer foil contained the TMAH in methanol, 34 nmol of 1-fluoronaphthalene, and 25 nmol of pyrene, both as recovery standards that would not react under thermochemolysis. The inner foil contained 13 nmol of nonanoic acid as the internal standard. The 1-fluoronaphthalene was detected in both GC1 and GC2 data. The detection of 1-fluoronaphthalene and trimethylamine (TMA) confirms that the cup was punctured and the reagent heated such that the TMAH decomposed to its primary byproducts and the recovery standard was released. The nonanoic acid internal standard should have been methylated by TMAH during thermochemolysis and eluted in GC1 at ~17.5 min and in GC2 at ~15.0 min. However, neither nonanoic acid nor its methyl ester was detected. SAM flight engineering data indicated puncture of the foils containing TMAH and the nonanoic acid. The lack of internal standard detection is therefore an artifact of the experimental design, as benchtop experiments confirm that the sampling cadence (the venting during EGA subsampling), driven by SAM valve configurations, led to the loss of the nonanoic acid. The pyrene recovery standard elutes outside of the GC1 heating window and GC2 stopped heating prematurely, before possible pyrene release to the detector; thus, the experiment did not enable pyrene detection.
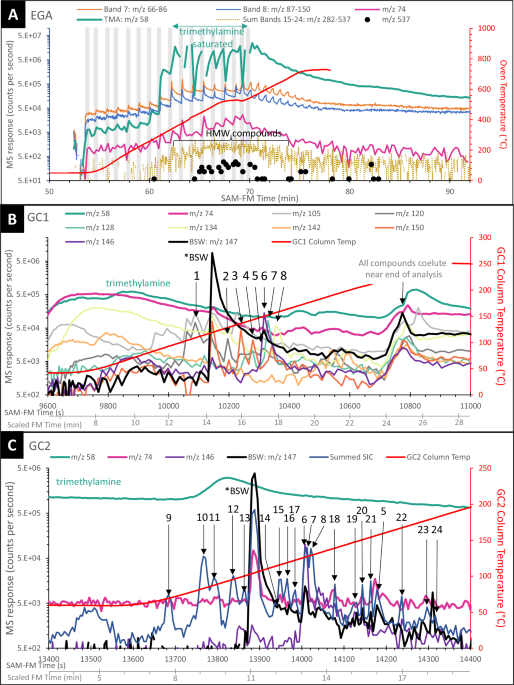
SAM-FM EGA results revealed high-molecular-weight (HMW) molecules with ions having mass-to-charge ratios (m/z) up to 537 (Fig. 1A) and a complex signal due to detector saturation with ions such as TMA (m/z 58), which is produced by the decomposition of TMAH. The sawtooth pattern is due to the subsampling process described above. EGA results for select m/z values are consistent with the presence of benzene, toluene, trimethyl- and tetramethylbenzene, naphthalene, and methylnaphthalene (Table S-1). As the gases from EGA do not interact with the HC trap, these m/z values are not related to any HC trap contamination (Fig. S-28). HMW molecules in both EGA and GC-MS signals (Fig. 1A–C) were detected as the total ion current generated by the sum of ion intervals (bands 15-24) over the m/z 282-537 range. Molecules were identified based both on corroborating National Institute of Standards and Technology (NIST) mass spectra17 comparisons and, when possible, with retention time (Rt) experiments using the SAM-like GC-MS breadboard (BB) with spare flight columns (i.e.,18,19).
Fig. 1: Representative traces extracted from the EGA analysis and chromatograms from the Gas Chromatograph 1 and Gas Chromatograph 2 columns from selected m/z values or bands covering a range of masses.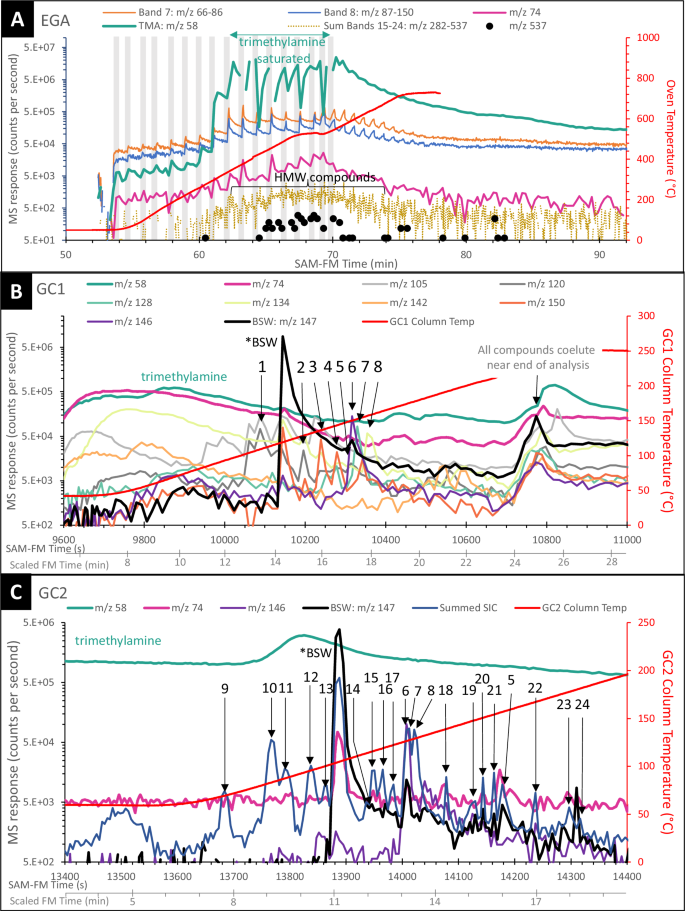 The alternative text for this image may have been generated using AI.
The alternative text for this image may have been generated using AI.
Gray boxes in A denote the range of time and oven temperature over which volatiles detected in EGA were subsampled for GC-MS analysis. m/z = 74, a trace mass for FAMEs, is included. Eight molecules are highlighted in GC1 in B, and 20 molecules are highlighted in GC2 in C. For GC2, summed SIC is the sum of selected ions m/z 121, 128, 130, 134, 135, 136, 140, 142, 144, 148, 149, 150, 161. Peak 1 – trimethylbenzene, Peak 2 – tetramethylbenzene, Peak 5 – derivatized dimethylsilanediol, Peak 6 – the recovery standard 1-fluoronaphthalene, Peak 7 – naphthalene, Peak 8 – benzothiophene, Peak 11 methyl benzoate, Peak 15 – dihydronaphthalene, Peak 21 – methylnaphthalene, Peak 23 – diphenylmethane. Peaks 3, 4, 9, 10, 12, 13, 14, 16, 17, 18, 19, 20, 22, and 24 are unidentified. Peak numbers listed correspond to peak identifications in Tables 1 and S1. SAM-FM Time in seconds reflects the serial data collection on GC1 and GC2. Scaled FM Time in minutes reflects the retention time scaled to each GC channel and corresponds to the SAM GC Breadboard retention time in Table S-1. EGA evolved gas analysis, GC gas chromatograph, MS mass spectrometry, FAMEs fatty acid methyl esters, SIC select ion chromatogram, SAM-FM Sample Analysis at Mars Flight Model, BSW bisilylated water, HMW high molecular weight, TMA trimethylamine.
Three GC-MS experiments were performed on the Mary Anning sample: neat pyrolysis (heating under He flow without additional reagents), TMAH thermochemolysis, and MTBSTFA derivatization. Seven molecules were detected in the SAM-FM TMAH GC-MS data for MA3 that were absent in the pre-sample-analysis and post-analysis clean-up (Fig. 2). These molecules are trimethylbenzene, tetramethylbenzene, methyl benzoate (benzoic acid methyl ester), dihydronaphthalene, naphthalene, benzothiophene, and methylnaphthalene (Table 1). These 7 molecules represent only a portion of the organics generated with this experiment, with 30 chromatographic peaks associated with one or more molecules. The identity of these remaining molecules cannot be confirmed with retention time analyzes, but we can speculate on their identity based on mass spectra. Of these peaks, only bisilylated water (BSW) is also present in the pre-sample-analysis and post-analysis clean-up (Fig. 2A, C).
Fig. 2: SAM-Flight Model identification of aromatic molecules compared with results from laboratory benchtop retention time experiments. The alternative text for this image may have been generated using AI.
The alternative text for this image may have been generated using AI.
SAM-Flight Model GC1 and GC2 chromatograms (A, C) and laboratory benchtop retention time experiments (B, D) on the SAM-Gas Chromatograph Breadboard confirm the presence of trimethylbenzene, tetramethylbenzene, methyl benzoate, dihydronaphthalene, naphthalene, and benzothiophene in the Mary Anning TMAH experiment. Methylnaphthalene is tenatively identified. Lines represent spectra summed from m/z 120, 121, 128, 130, 134, 135, 136, 140, 142, 144, 148, 149, and 150. In A, C, solid blue line is the TMAH experiment, dashed orange line is the pre-sample-analysis performed prior to the TMAH experiment (same conditions as the experiment but without a sample or TMAH), dotted gray line is the post-TMAH experiment clean up analysis. In B, D, black and orange lines represent molecular standards analyzed on the relevant SAM-GC Breadboard GC columns. Numbers above peaks correspond to peak numbers in Fig. 1 and Table 1. GC gas chromatograph, TMAH tetramethylammonium hydroxide, BSW bisilylated water.
Table 1 Seven confirmed molecular identities in SAM-Flight Model Gas Chromatograph 1 and Gas Chromatograph 2 data
MTBSTFA and its reaction products are present within the SAM-SMS. The presence of BSW, among other products, is due to a MTBSTFA cup leak detected since Curiosity’s landing20 and two subsequent full-cup MTBSTFA derivatization experiments (on sols 1909 and 2885)6,7. A second known SAM-internal source of organic molecules in GC-MS includes thermal degradation products (primarily benzene and toluene) of Tenax TA adsorbent in the hydrocarbon trap generated every heating cycle used to release trapped sample analytes21. MTBSTFA-leak and trap derived molecules can complicate interpretations to discern the source of organics detected in SAM, however those byproducts are well characterized21.
Organics detected in GC1
Four molecules detected in GC1 (Table 1) are confirmed as trimethylbenzene, tetramethylbenzene, naphthalene, and benzothiophene (peaks 1, 2, 7, and 8, respectively, Fig. 2B). Mass spectra corresponding to two additional molecules were identified in GC1 data, but laboratory retention time experiments on a SAM GC-MS BB did not confirm these identifications. Regardless, the compelling nature of the mass spectra leads us to report on them here as plausible detections of molecules with very similar fragmentation patterns. These detections include a benzene ring with N- and/or O-bearing functional groups (Peak 3) and a benzene ring with N-, COOH-, and/or CH3- functional groups (Peak 4).
At ~25 min Rt and a column temperature of ~220 °C, a complex co-elution occurs in GC1 (Fig. 1B) which may be due to a column saturation effect. The high flux of BSW and TMA is expected to lead to a column saturation effect that does not permit the full portion of these compounds to efficiently adsorb into the column film. These compounds are initially trapped but then elute at high temperature along with other HMW molecules and the expected column bleed.
Organics detected in GC2
More than 20 discrete peaks were present in the GC2 chromatogram (Table 1). Confirmed molecules include methyl benzoate, dihydronaphthalene, naphthalene, benzothiophene, and methylnaphthalene (peaks 11, 15, 7, 8, and 21, respectively).
Sixteen peaks remain unidentified, but similarities in mass spectral fragments can yield insights into likely molecular identities22. Identifications include a methylated benzene ring with an amine functional group (Peaks 9, 10, and 17), a multiply-methylated benzene ring with alcohol or methoxy functional groups (Peak 12), a benzene ring with methyl, ethyl, and/or isopropyl groups (Peak 13), a single ring aromatic with methyl and/or ethyl functional groups (Peak 14), a single ring aromatic with methoxy, alcohol, isopropyl and/or methyl functional groups (Peak 16), a single ring aromatic with alcohol, amine, and/or methyl functional groups (Peak 18), a dicyclic aromatic with a methyl group (Peak 19), a multiply-methylated benzene ring (Peak 20), and a methoxy-bearing dicyclic aromatic (Peak 24). Peak 22 is characterized by ions with m/z 145, 144, and 146. Comparisons with the NIST library yielded a best match with dimethyl-indole, with the spectral comparison especially compelling (Fig. S-24). However, the retention time of dimethyl-indole on the SAM-like GC-MS BB GC2 column was offset at 20.7 min relative to the candidate molecule at 17.0 min. We therefore suggest this molecule consists of a methylated dicyclic aromatic with a N-heterocycle.
Highly volatile molecules detected in GC1 separations were likely lost from GC2, which warmed as adjacent GC1 heated, volatilizing some of the passively retained analytes at GC2’s front end. Short-chained carboxylic acids likely passed through GC2 undetected before the start of the GC2 measurement. The carbon chains of saturated carboxylic acid methyl esters (CAMEs) that should elute within the time range of GC2 are C4 through C11; C4 through ~C16 would theoretically elute in the GC1 range. No CAMEs, including the C9 internal standard, were detected in the TMAH experiment.
Complimentary laboratory experiments
As a geochemical comparison, Murchison meteorite and other carbonaceous chondrites can be used as a reference for exogenous organic matter delivered to Mars23. To help interpret SAM data and the potential origin for organics detected, benchtop laboratory experiments were performed on the Murchison meteorite. During TMAH thermochemolysis, the Murchison meteorite insoluble organic matter (IOM)24 is degraded into its constituent parts, some of which are detectable with GC-MS5,23. These benchtop experiments included neat pyrolysis and TMAH thermochemolysis with and without a small contribution of MTBSTFA vapor to mimic the MTBSTA leak in SAM. Experiments were performed with both flash pyrolysis at 600 °C and the SAM pyrolysis ramp of 35 °C/min.
These benchtop experiments on the Murchison meteorite liberated a variety of aromatic acids and hydrocarbons, aliphatic carboxylic acids, and organosulfur compounds. Sixteen of the 28 species confirmed or tentatively identified in the SAM-FM TMAH experiment are also identified in the TMAH thermochemolysis of the Murchison meteorite. Molecules detected with neat pyrolysis and/or TMAH thermochemolysis of Murchison include benzene, toluene, tri-, tetra-, and pentamethylbenzene, trimethylbenzenamine, ethyltetramethylcyclopentadiene(?), naphthalene, methyl benzoate, benzothiophene, methylnaphthalene, dimethyl-indole, methoxybenzene, and methoxynaphthalene23,25 (Table S-1).
These benchtop experiments demonstrated that, with flash TMAH thermochemolysis at 600 °C, aromatic acids are generated from cleaved ester aliphatic linkages joining the IOM macromolecular framework in the Murchison meteorite24. These acids could then be further decarboxylated under the pyrolysis conditions, yielding benzene and naphthalene. However, under SAM-like pyrolysis conditions, those aromatic acids (e.g., benzoic acid) that might have decarboxylated into an alkylbenzene would instead be methylated into volatile, thermally stable methyl ester derivatives24. Both products (e.g., aromatic acids and their decarboxylated alkyl-aromatic derivatives) are detected in the SAM-FM TMAH experiment.
Benchtop TMAH experiments yielded methylated and non-methylated species, indicating partial methylation of compounds from decreased TMAH thermochemolysis reaction rates at the slower SAM pyrolysis ramp. This finding is expected as TMAH derivatives from the SAM-like ramp experiment displayed reduced relative abundances, ca. two orders of magnitude lower as compared to flash conditions. Because of this, chromatograms from benchtop SAM-like TMAH thermochemolysis experiments contain a combination of peaks representing compounds detected in pyrolysis experiments with and without TMAH. Methylation of free benzene or naphthalene with TMAH is unlikely, as the reagent hydrolyzes labile H from polar functional groups which are lacking on these aromatic molecules. Direct C-methylation of naphthalene with TMAH was also not observed in benchtop experiments26. These results indicate that the methylated benzene and naphthalene derivatives in the SAM-FM TMAH experiment are derived from a macromolecular source like the Murchison macromolecular carbon.
Detection of methyl benzoate
Methyl benzoate was detected in the SAM-FM TMAH experiment in GC2. Benzoic acid has been detected previously with SAM from both Mars-indigenous3,7,27 and SAM-internal7,21 sources. SAM-internal sources are detailed in refs. 7,19. Previous work has documented that benzoic acid may be produced through the oxidation of organic molecules present in the presence of perchlorates in the martian regolith and/or the thermal decomposition of the Tenax TA adsorbent present in the SAM trap(s). Although reactions of TMAH with 10 wt% CaClO4 during benchtop flash pyrolysis experiments28 have been shown to generate methyl benzoate amongst other products (but not naphthalene), this is not a potential source for the SAM-FM TMAH experiment. Perchlorate was not detected in the Mary Anning target, and regardless, average perchlorate concentrations in Martian regolith are far lower at 0.5 to 1.0 wt%. Benzoic acid and naphthalene have also been detected after overheating (>500 °C) and repeated activations of Tenax during benchtop experiments21. However, the SAM-FM Tenax has not been overheated.
Benzoic acid was postulated to be the preferred precursor for the chlorobenzene that was detected by SAM and demonstrated to be indigenous in the Cumberland mudstone at the base of Aeolis Mons3,27. Notably, chlorobenzene was detected at background levels in the Mary Anning neat pyrolysis experiment6. Chlorobenzene can be produced by reactions between benzoic acid and perchlorate at elevated temperatures27, but again, perchlorates were not detected at Mary Anning. A portion of the benzoic acid derivatives generated in the TMAH and MTBSTFA6 experiments at Mary Anning may still be related to the decomposition of Tenax in the HC trap. However, its non-detection in the neat pyrolysis of MA, where similar analytical conditions were applied, indicates at least a partial martian origin for benzoic acid.
The Murchison meteorite benchtop experiments demonstrated that pyrolysis-only treatments could generate benzoic acid, but only with TMAH thermochemolysis does methyl benzoate form, even without the use of a hydrocarbon trap23. The presence of methyl benzoate in the Mary Anning TMAH experiment confirms that TMAH successfully reacted with the Martian sample to generate a CAME.
Detection of benzothiophene
A variety of sulfur-bearing molecules have been previously detected on Mars with SAM (i.e.,3,5,6), including dithiapentane, dithiolane, and trithiane, which were detected in the neat pyrolysis GC-MS analysis of Mary Anning. The benzothiophene detection reported here is robust, detected in both GC1 and GC2, and the first confirmation of benzothiophene on Mars, as the molecule was only weakly indicated in lacustrine sediments in the Gale crater floor5, and was lacking from the MA neat pyrolysis experiment. These S-bearing molecules were not methylated but could have been liberated from a macromolecular source by TMAH thermochemolysis. No known pyrolysis or thermochemolysis process on SAM would generate benzothiophene as a SAM-byproduct, but it is prevalent in carbonaceous meteorites (see ref.29 and references therein) and readily liberated by pyrolysis and TMAH thermochemolysis from carbonaceous meteorites such as Murchison23, as well as found in-situ and confirmed by extraction in the Tissint meteorite30,31. Sulfurization has been invoked as a preservation mechanism for other S-bearing organics identified on Mars5. Together, these findings suggest that the benzothiophene in Mary Anning was liberated from an indigenous macromolecular source.
Detection of methylated cyclic molecules
Methylated benzene and naphthalene products were detected, which suggests that a larger macromolecular structure was broken apart by the TMAH thermochemolysis process. The methylated benzene and naphthalene derivatives are potentially sourced from partial methylation of aromatic acids such as benzoic and naphthoic acids. Although C3 and C4 methylated benzene and C1 and C2 methylated naphthalene derivatives were generated with benchtop experiments on Murchison meteorite with SAM-ramp neat pyrolysis and TMAH thermochemolysis, methylated benzene and naphthalene derivatives were not generated in the Mary Anning neat pyrolysis experiment6. We note that none of the organic molecules identified in the TMAH thermochemolysis experiment at Mary Anning were also present in the Mary Anning neat pyrolysis experiment, indicating unique experimental conditions that yielded different organic molecules between the two experiments. There are more similarities between the Mary Anning MTBSTFA experiment and the TMAH experiment than there are between the TMAH experiment and the neat pyrolysis experiment, including the presence (or presence at background levels) of benzoic acid (in the t-BDMS or methyl ester form), diphenylmethane/methylbiphenol (considered a SAM internal source), and naphthalene (potentially indigenous)6.
Detection of nitrogen-bearing molecules
The mass spectra for peak 22 in the TMAH experiment are most consistent with the N-heterocycle dimethyl-indole. The most similar molecules identified in benchtop TMAH thermochemolysis experiments with Murchison meteorite yielded di-, tri- and tetramethyl-indole, and 1-methyl-pyrrole23. Though several N-heterocycles have been identified in carbonaceous chondrites after aqueous or acid extraction32,33,34,35 the same cannot be said for the martian meteorites, where non-terrestrial amino acids were detected in RBT 04262. N-heterocycles were not detected by the same technique in ALH84001, ALHA77005, EETA79001, or MIL0334636, but have been confirmed in Tissint, Nakhla, and NWA195030. We suggest that the molecule present at peak 22 consists of a methylated double-ring aromatic with a N-heterocycle. This is an exciting possibility, as N-heterocycles are fundamental components of astrobiologically relevant molecules, such as nucleic acids.
Molecules containing nitrogen heteroatoms were also detected in GC1 and/or GC2 data at peaks 9, 10, and 17 (potentially trimethyl-benzenamine, dimethyl-benzenamine, and tetramethyl-benzenamine, respectively)37. Similar molecules such as N,N,2-trimethyl-benzenamine and N,N-dimethyl-benzenemethanamine are identified in the benchtop neat pyrolysis and TMAH experiments of the Murchison meteorite23,38. Several TMAH thermochemolysis products in these benchtop experiments did yield other molecules bearing dimethylamine functional groups, such as methyl 3-dimethylaminopropionate and N,N,N’,N’-tetramethyl-1,2-ethanediamine23. Additionally, aliphatic amines have been detected in hydrolyzed water extracts of Murchison39,40,41. Furthermore, nitrogen functionality and the presence of aliphatic and aromatic nitrogen species have been determined from the Tissint meteorite31.
Benzonitrile is another common pyrolysis product of some meteorites, and has been found in the Murchison meteorite23 and in the martian meteorite Nakhla40 with its Martian origin confirmed30. Benzonitrile was identified in the Mary Anning MTBSTFA experiment, and in other Glen Torridon drill targets (Groken and Nontron)6, but was not identified from the SAM TMAH experiment. The three peaks associated with N-bearing organics indicated in this work may yet be products of reactions between TMAH and a macromolecular precursor in Mary Anning, although reactions between TMAH and the SAM hydrocarbon trap cannot be totally discounted. TMAH produces a variety of methylated amine products due to the methylamine component of the organic salt. During thermochemolysis, intermediate products of both TMAH and sample organics could react to form a greater variety of N-bearing products.
Detection of oxygen-bearing molecules
Oxygen-bearing molecules indicated in the SAM-FM TMAH experiment are comparable to those identified in benchtop TMAH thermochemolysis of Murchison, such as methoxymethyl-benzene and anisole23. Phenol is the most abundant O-containing organic compound liberated from the Murchison meteorite using neat pyrolysis. With TMAH thermochemolysis (under 600 °C flash benchtop conditions), phenol is converted to anisole (methoxybenzene). Under SAM ramp TMAH conditions, both phenol and anisole are detected. In the SAM-FM TMAH data, it can be challenging to differentiate between these similar molecules. For example, peaks 12 and 16 are likely “a benzene ring with alcohol” and both species are reflected in the closest NIST matches (Figs. S11 and S15). Peak 24 is likely a “methoxy-bearing double ring aromatic” and the closest NIST match is methoxynaphthalene (which was also identified in the Murchison TMAH experiments, 23). Peak 18 is likely “a single ring aromatic with alcohol and/or methyl functional groups” and the closest NIST match is tetramethyl-phenol. The detection of both species is consistent with the partial methylation of likely meteoritic macromolecular carbon due to decreased TMAH thermochemolysis reaction rates at the slower SAM pyrolysis ramp. The observation that more of these species were observed in the newly commissioned GC2 column, which has a stationary film with lower (5%) phenyl moieties than GC1 (20%), supports the conclusion that these species are not artifacts from column bleed. Column degradation should yield at most a minor contribution to the molecules identified from this column, although hydrocarbon trap products may still be a partial source of the benzoic acid (see Supplemental Information) discussed above.
Evolved gas analysis
All molecules detected in the SAM-FM TMAH experiment are cyclic. EGA results for select m/z values are consistent with the presence of benzene, toluene, tri- and tetramethylbenzene, naphthalene, and methylnaphthalene (Table S-1), and these are consistent with species identified in benchtop TMAH thermochemolysis experiments of the Murchison meteorite23. Molecular signals in EGA cannot represent components produced on the SAM traps as the gas flow is diverted directly into the MS without encountering the HC trap. Benzene, toluene, alkylbenzenes and traces of naphthalene and benzothiophene signals above blanks levels were present in SAM-FM EGA of pyrolysis products from ancient Martian lacustrine sediments at the base of Aeolis Mons5. The study here now confirms the detection of naphthalene and benzothiophene with GC-MS.
Lack of aliphatic carboxylic acids
One class of molecules commonly liberated from carbonaceous meteorites via TMAH thermochemolysis is CAMEs. TMAH methylates carboxylic acids with high efficiency42 and CAMEs have been generated from benchtop TMAH thermochemolysis of Murchison meteorite23. Benzoic acid in the form of methyl benzoate was the only carboxylic acid detected in the SAM-FM TMAH experiment. The lack of aliphatic CAMEs in these data suggests that (1) CAMEs are not present in detectable amounts with SAM and/or (2) flight operating conditions require optimization before they can be definitively detected, with the latter a preferred explanation as described above.
Mars-indigenous sources of organics
This study documents the first in situ TMAH wet chemistry experiment on Mars, which was performed by the SAM-FM instrument aboard the Curiosity rover. The TMAH experiment was successful based on confirmed puncture of the foils on the TMAH cup, detection of the recovery standard 1-fluoronaphthalene, and presence of the TMAH decomposition product trimethylamine. The detection of methyl benzoate confirms that TMAH reacted to form this methyl ester product. This study has identified over 20 aromatic and cyclic molecules with methyl and ester/carboxylic acid functional groups, and sulfur-, oxygen-, and nitrogen-bearing organics, including the first detection of a possible N-heterocycle. Both one- and two-ring aromatics have been identified, including the first confirmation of both naphthalene and benzothiophene. Benzothiophene is a known component of meteoritic macromolecular carbon and represents the largest confirmed underivatized aromatic molecule identified thus far as indigenous to Mars. Molecular abundances in the SAM TMAH experiment range from 0.1 ± 0.0 to 1.7 ± 0.3 nmol, consistent with the range of abundances of individual molecules identified by SAM from other Gale crater outcrops3,6. The detection of single and dicyclic aromatics and macromolecular carbon in near-surface Mars outcrops is consistent with the findings from analyzes of martian meteorites30,37,43 and the NASA Mars 2020 Perseverance rover44,45,46. The detection of 1–2 ring aromatics in the SAM TMAH experiment, but not in neat pyrolysis, suggests that these molecules derive from a macromolecular source that was cleaved by TMAH thermochemolysis.
We propose that this suite of organics represents TMAH thermochemolysis breakdown products from ancient organic macromolecular material that has been preserved in billions-of-years-old sedimentary rocks in Gale crater. Analysis of the spatial distribution of the organic matter is not possible with SAM, therefore the origin of this material as being introduced from meteorites, abiotically produced via aqueous processing such as serpentinization or electrochemical production, is currently unknown22,30. Regardless, confirmation of macromolecular organic matter supports the possibility that future optimized TMAH thermochemolysis experiments can liberate ancient biosignatures preserved in macromolecules on Mars (if present). The broad structural variety of organic molecules observed in situ from surface materials suggests some chemical diversity is preserved in ancient Martian sediments despite >3.5 billion years of diagenesis and radiation exposure. These results expand the library of confirmed and suggested organic molecules preserved over deep geologic time in the Martian near-surface and confirm the presence of macromolecular carbon on Mars.